INSIGHT
The vaccines for COVID-19 produced by Pfizer-BioNTech and Moderna were the first mRNA vaccines authorized for use in the general public. mRNA vaccines were a long-standing ambition for scientists, as it was hoped that they would offer many advantages over traditional vaccines[1]. However, there were significant technical challenges that took decades to overcome.
How do mRNA vaccines work?
Vaccines work by mimicking an infection and training the immune system to recognize a disease-causing agent, such as viruses or bacteria. This training allows the body to respond more rapidly when it encounters that virus or bacteria in the future. This is usually done by introducing a weakened or inactivated version of the pathogen or parts from it, such as proteins, into the body.
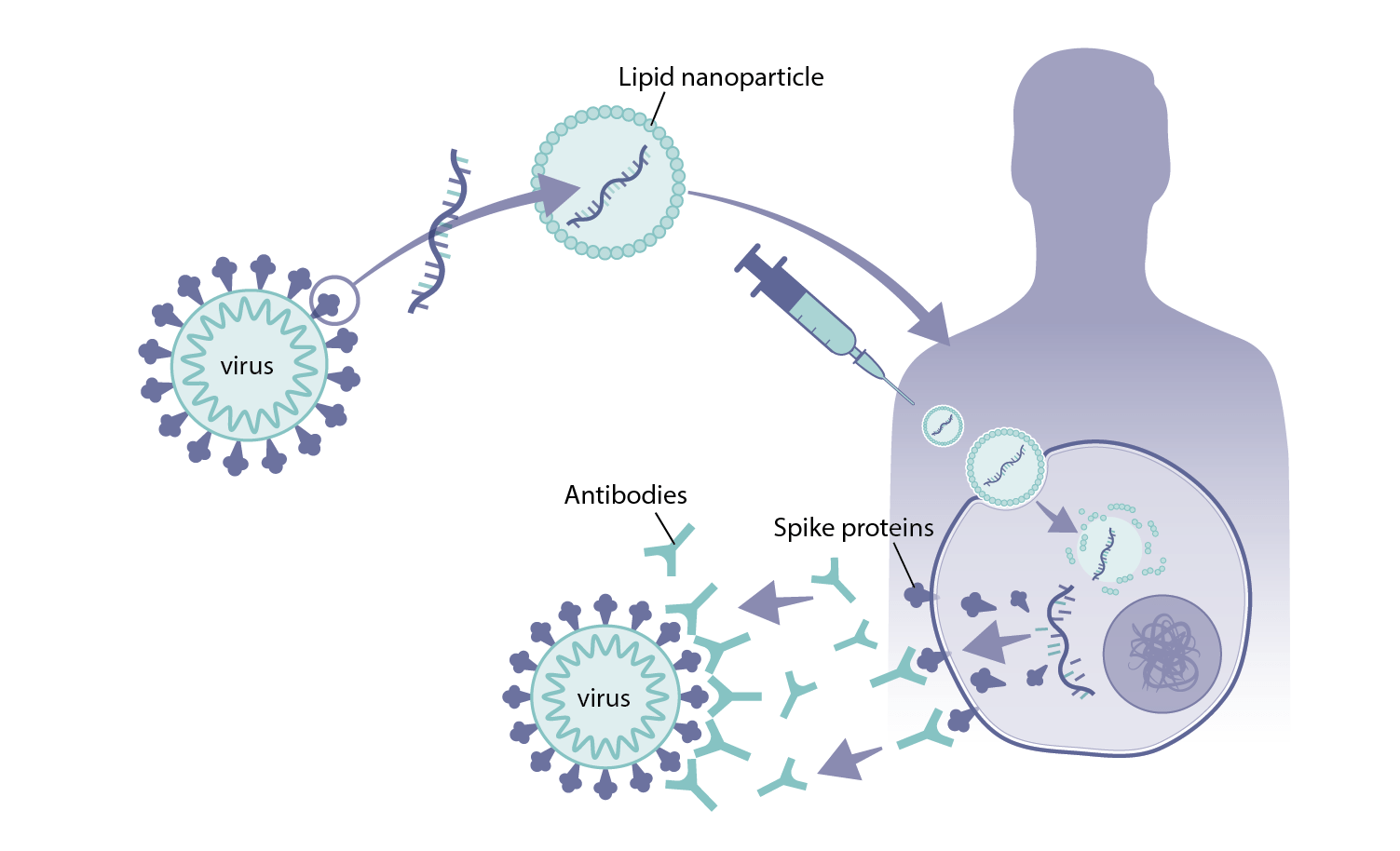
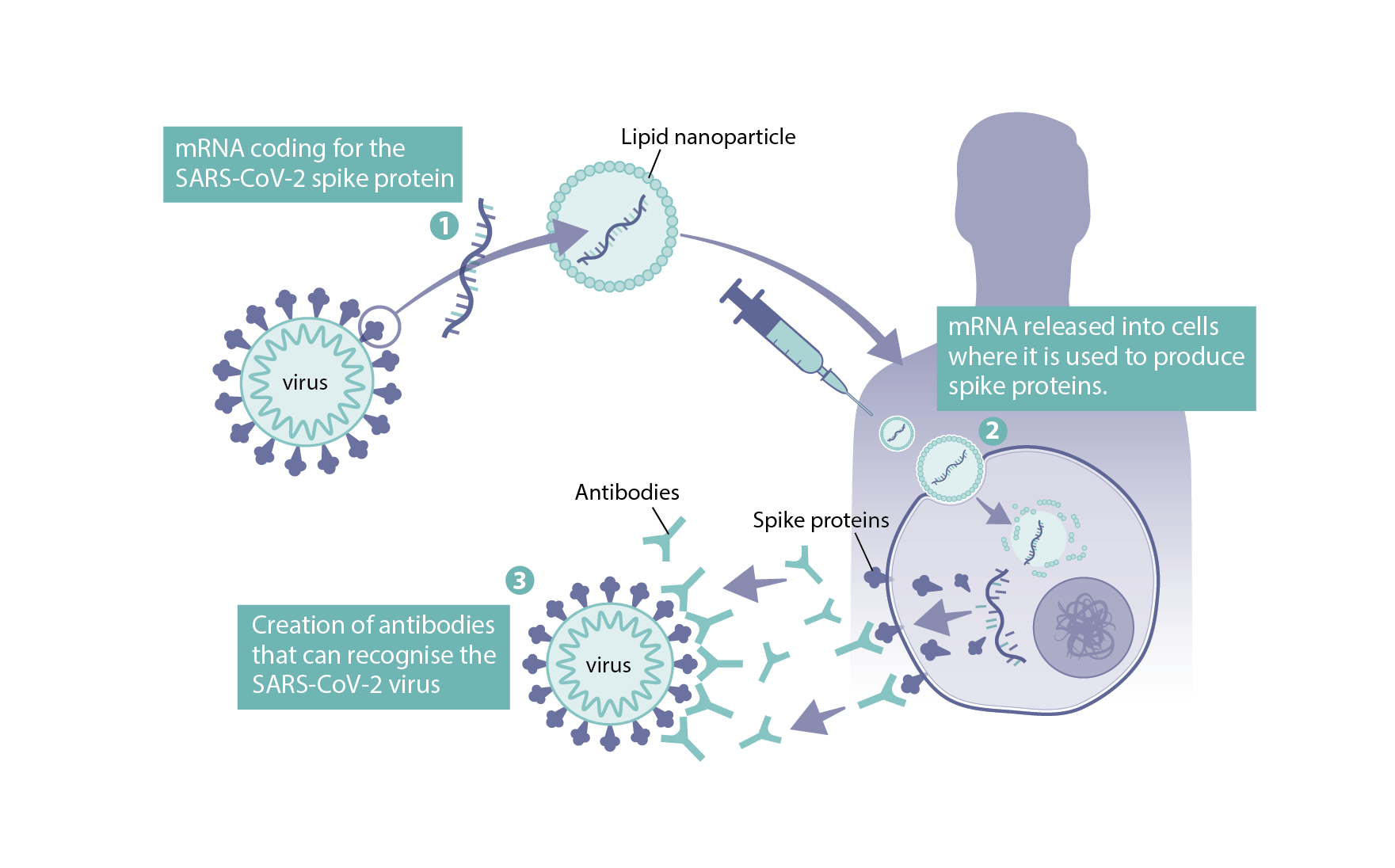
mRNA vaccines take a different approach. Instead of including a protein from the pathogen or the pathogen itself in the vaccine, they contain the genetic instructions for producing that protein in our own cells. These genetic instructions, known as messenger RNA (ribonucleic acid), are what our cells use as part of the normal machinery to assemble proteins that are needed to keep the cell alive.
Once the cell receives these genetic instructions, it produces the protein from the virus that can then be used to train the immune system. The mRNA genetic instructions are destroyed by the cell as part of this process.
After overcoming the technical challenge of getting mRNA to the cell, scientists hope mRNA vaccines could save lives from a wide range of diseases. – Illustration by Bruno Bourgeois, for Health Feedback
What are the advantages of mRNA vaccines?
Because mRNA vaccines do not involve any viruses, they can be produced in a facility quicker and cheaper than conventional vaccines that require the viruses to be grown in cells or chicken eggs. This means that the production is easier to standardize and quicker to roll out for emerging outbreaks.
mRNA vaccines can be created for a new disease based on its genetic sequence. The mRNA can be synthesized in the lab quickly, meaning that a candidate vaccine can be ready for testing in the lab within days, often described as a ‘plug-and-play’ system. The speed of the mRNA vaccine production could offer an advantage for rapidly changing diseases, such as influenza. The current system for influenza requires predicting the most common strains months ahead of flu season. If the wrong strains are chosen, the vaccine is less effective. mRNA vaccines could shorten this timeframe and allow easier adjustment to different strains.
“It’s really easy to manipulate the coding sequence of the vaccine to try new hypotheses and strategies that may make for better vaccines,” said Justin Richner, a vaccinologist at the University of Illinois College of Medicine in Chicago, who is developing an mRNA-based dengue vaccine
Conventional vaccine technology has been unable to rapidly develop effective protection against a number of diseases, including HIV and Ebola[1]. mRNA vaccines offer a new approach that may have more success against challenging diseases. Tests in animals have shown that the technology can stimulate a strong immune response and are effective against Zika, rabies, and influenza[1].
Why didn’t we have mRNA vaccines earlier?
Efforts to create an mRNA vaccine have been ongoing since 1993, when scientists tested mRNA for influenza in mice[2]. However, there were many technical hurdles that needed to be overcome before they could be tested in people.
One challenge is that messenger mRNA itself could also potentially cause an unintended immune reaction. In 2005, scientists found a way to alter the mRNA to make it similar to what is naturally found in mammals, reducing the immune response and increasing protein production[3].
The other major hurdle to overcome with mRNA vaccines is the instability of mRNA. mRNA is quickly broken down in the body, so scientists needed to develop technologies to protect it long enough to reach the cell. One way of doing this is to trap the mRNA in fat droplets, known as lipid nanoparticles. This technology, first tested in vaccines in 2012, allows the mRNA to reach and enter the cell before being released[4].
In 2012, the US Defense Advanced Research Projects Agency began funding research at major pharmaceutical companies to develop the technology. However, many of these companies did not continue with the research apparently due to concern over the commercial uncertainty of investing in new technology that had not yet been approved. Dan Wattendorf, a former program manager at DARPA, said to Nature, “They were reticent about taking on any risk with a new regulatory pathway for vaccines, even though the data looked good.”
Some smaller biotech companies, such as Moderna and CureVac, continued to develop mRNA vaccines. Human testing of mRNA vaccines began in 2013 with CureVac’s rabies vaccine[5]. At the beginning of 2020, only a dozen mRNA vaccine candidates had been tested in people. Although some of these showed good safety and immune response[5,6], only one, for cytomegalovirus, had yet gone beyond initial safety testing.
Although no mRNA vaccines had been approved by the start of 2020, the technology platform for creating new vaccines was established. That meant it would be relatively straightforward to create a vaccine for a new pathogen based on its genetic sequence.
How were mRNA vaccines developed for COVID-19?
Companies that had been working on mRNA vaccines for years, such as Moderna and BioNTech, were able to create an mRNA vaccine when COVID-19 began to spread worldwide. Because the technology only requires the genetic sequence of the virus rather than a sample of the virus itself, they could begin work quickly.
The vaccines use a modified form of the genetic sequence for the virus’s spike protein. Research on previous outbreaks of similar coronaviruses, SARS and MERS, suggested that this would trigger the best immune response to the SARS-CoV-2 virus. The spike protein is modified so that it holds the structure that would be found on the virus before it binds to the host cell.
The vaccines were closely monitored during clinical trials in thousands of people for safety and effectiveness. Both the vaccines from Moderna and Pfizer-BioNTech were over 90% effective in preventing symptomatic cases of COVID-19 in clinical trials.
Thanks to the ‘plug-and-play’ technology behind mRNA vaccines, as well as record investment, the vaccines for COVID-19 were developed faster than any other vaccine.
What are the side effects of the COVID-19 mRNA vaccines?
The clinical trials of the COVID-19 mRNA vaccines showed that there are common side effects, including headaches, muscle pain, and fever. These are more common after the second dose of the vaccine and in younger people. These types of side effects are typical of the immune response expected from a vaccine.
When the vaccines were rolled out to the general public, rare cases of anaphylaxis were reported. These were estimated to occur in 2.5 cases per million Moderna vaccines and 11 cases per million Pfizer vaccines. This is believed to be due to reactions against one of the ingredients in the mRNA vaccines, polyethylene glycol[7,8]. The U.S. Centers for Disease Control and Prevention has advised that anyone with a history of allergic reactions to polyethylene glycol should be offered an alternative to the mRNA vaccines, such as the vaccine produced by Johnson & Johnson.
What ingredients are in the vaccines?
The vaccine contains mRNA encapsulated in lipid nanoparticles. The mRNA encodes for the full-length SARS-CoV-2 spike protein with some minor changes for stability.
The primary ingredients in Moderna and Pfizer vaccines are the mRNA, lipids, acidity regulators, salts, sucrose (a sugar), and water. The lipids form the nanoparticle or bubble to protect the mRNA until it reaches the cell. Polyethylene glycol is attached to the lipids to prevent the particles from sticking together and being detected by the immune system. The acidity regulators, or buffers, keep the pH of the solution at the right level for the body. The sucrose protects the nanoparticles when they are frozen and prevents them from sticking together.
What misinformation is being shared about mRNA vaccines?
The unfamiliarity of mRNA vaccines among the general public led to a significant level of misinformation spread online.
In particular, the use of mRNA as a form of genetic material was misrepresented on social media posts that suggest the vaccines can change our DNA. One claim stated that the vaccine would “permanently alter your DNA” and another said that the spike protein produced by the vaccine would “bind to your DNA and irreversibly change it”. As has been covered in reviews on Health Feedback for these claims, neither the mRNA nor the spike protein enters the nucleus of the cell where the DNA is stored. They do not come into contact with our DNA or alter it in any way.
Similarly, the approach of the vaccines using our cellular machinery to produce the SARS-CoV-2 spike protein has been misunderstood. The mRNA introduced through the vaccine survives just long enough to allow the cells to produce spike protein for generating an immune response. Health Feedback has debunked claims that the vaccines will produce more viruses or promote cancer.
There were also claims that vaccinated people shed the spike protein, which could then cause an infection in unvaccinated people nearby. However, it is not possible for the spike protein to cause an infection, that would require a fully-assembled, functional virus. Other claims suggested that mRNA COVID-19 vaccines could cause neurodegenerative diseases based on a flawed speculative publication that was criticized by scientists.
Misinformation has also been shared concerning the safety of the lipid nanoparticles. Lipids have been used to package drug molecules for over twenty years to make the treatments more effective[9]. While they have not been used in previous vaccines, they have gone through thorough safety testing as part of the clinical trials. The cases of anaphylaxis, which are suspected to be linked to the lipids, are very rare.
There have even been claims that mRNA vaccines do not meet the definition of a vaccine. The CDC defines a vaccine as ‘a product that stimulates a person’s immune system to produce immunity to a specific disease, protecting the person from that disease.’ Although mRNA vaccines stimulate the immune system in a different way to traditional vaccines, they fully meet this definition.
REFERENCES
- 1 – Pardi et al. (2018) mRNA vaccines — a new era in vaccinology. Nature Reviews Drug Discovery.
- 2 – Martinon et al. (1993) Induction of virus-specific cytotoxic T lymphocytes in vivo by liposome-entrapped mRNA. European Journal of Immunology.
- 3 – Kariko et al. (2005) Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity.
- 4 – Geall et al. (2012) Nonviral delivery of self-amplifying RNA vaccines. Proceedings of the National Academy of Science.
- 5 – Alberer et al. (2017) Safety and immunogenicity of a mRNA rabies vaccine in healthy adults: an open-label, non-randomised, prospective, first-in-human phase 1 clinical trial. The Lancet.
- 6 – Feldman et al. (2019) mRNA vaccines against H10N8 and H7N9 influenza viruses of pandemic potential are immunogenic and well tolerated in healthy adults in phase 1 randomized clinical trials. Vaccine.
- 7 – Garvey and Nasser (2021) Anaphylaxis to the first COVID-19 vaccine: is polyethylene glycol (PEG) the culprit? British Journal of Anaesthesia.
- 8 – Sellaturay et al. (2021) Polyethylene glycol (PEG) is a cause of anaphylaxis to the Pfizer/BioNTech mRNA COVID‐19 vaccine. Clinical and experimental allergy.
- 9 – Boswell et al. (1998) AmBisome (liposomal amphotericin B): a comparative review. Journal of Clinical Pharmacology.
