- Health
No clinical evidence supports using cannabis to treat cancer or other conditions, although specific chemicals found in cannabis can be medically useful
Key takeaway
Despite growing interest in cannabis as an alternative therapy for various diseases, as well as promising results from pre-clinical studies on the efficacy of substances present in cannabis for treating cancer, there isn’t clinical evidence to support using cannabis as medical treatment. On the other hand, the purified or synthetic forms of THC and CBD, which are specific chemicals found in cannabis, have indeed been shown to be effective in the treatment of diseases. The U.S. FDA has approved drugs containing these substances.
Reviewed content
Verdict:
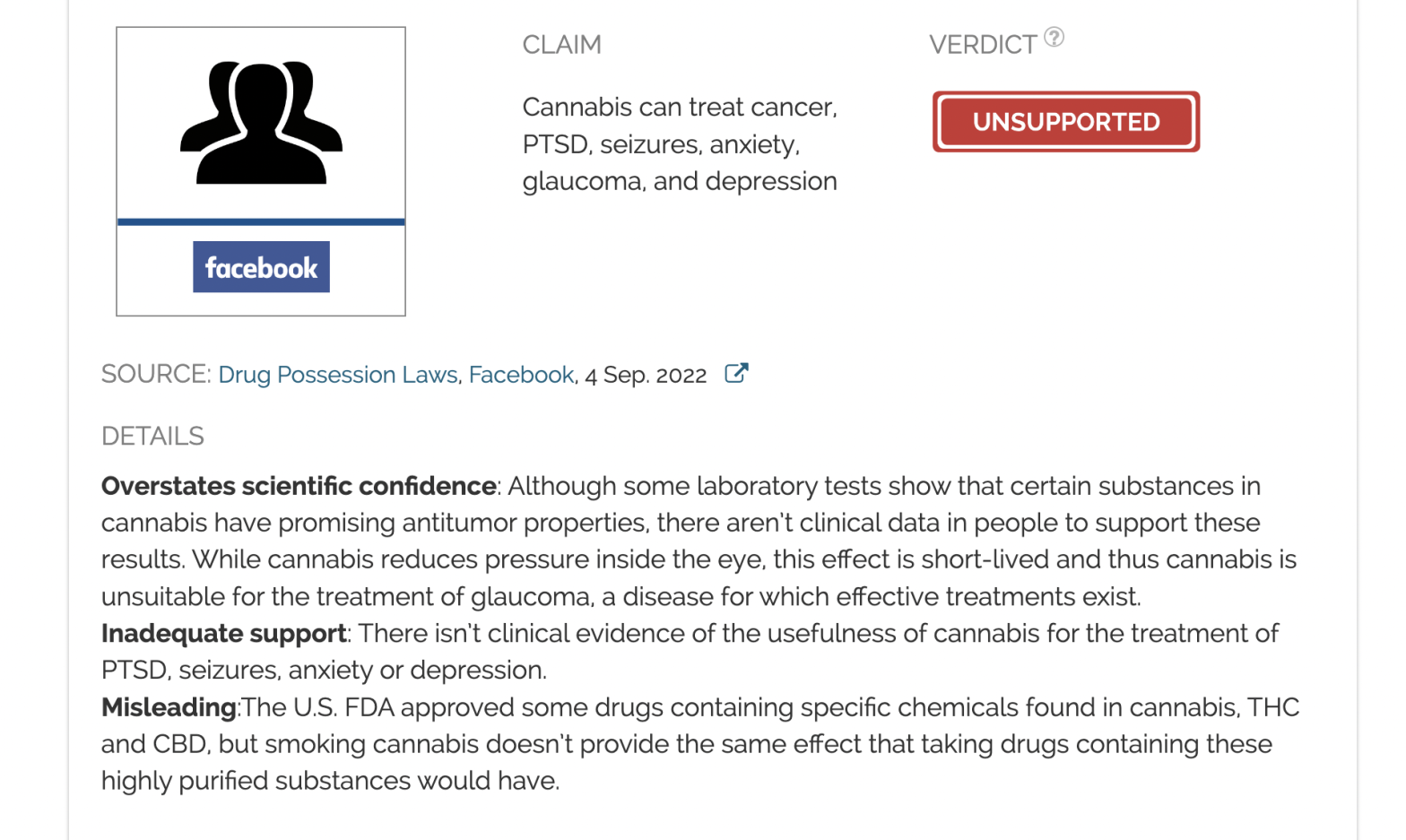
Claim:
Cannabis can treat cancer, PTSD, seizures, anxiety, glaucoma, and depression
Verdict detail
Overstates scientific confidence: Although some laboratory tests show that certain substances in cannabis have promising antitumor properties, there aren’t clinical data in people to support these results. While cannabis reduces pressure inside the eye, this effect is short-lived and thus cannabis is unsuitable for the treatment of glaucoma, a disease for which effective treatments exist.
Inadequate support: There isn’t clinical evidence of the usefulness of cannabis for the treatment of PTSD, seizures, anxiety or depression.
Misleading: The U.S. FDA approved some drugs containing specific chemicals found in cannabis, THC and CBD, but smoking cannabis doesn’t provide the same effect that taking drugs containing these highly purified substances would have.
Full Claim
“Cannabis is illegal because it can treat cancer, PTSD, seizures, anxiety, glaucoma, and depression, which means no money for pharmaceutical companies.”
Review
Cannabis has been proposed as an alternative therapy for various diseases, notably as a treatment for cancer. Such claims often go viral on social media, like this meme posted on the Facebook page Drug Possession Laws on 4 September 2022. The meme claimed that cannabis can treat cancer, post-traumatic stress disorder, seizures, anxiety, glaucoma, and depression. However, as we will explain in this article, there isn’t clinical evidence supporting most of these uses, and the claim is also potentially misleading, because it seems to conflate the cannabis plant with purified forms of specific chemicals in cannabis.
Cannabis, also known as marijuana, is a psychoactive drug obtained from plants of the genus Cannabis. Cannabis contains several substances that interact with the endocrine system, known as cannabinoids. The most notable cannabinoids are tetrahydrocannabinol (THC) and cannabidiol (CBD). These substances are present in cannabis and can also be produced synthetically.
Numerous pre-clinical and clinical studies have been conducted with cannabis or its compounds (mainly THC and CBD) to test its efficacy as a treatment for several diseases. Common limitations in several of these clinical studies are the small number of participants and short follow-up times, as we will show below with a few examples. To date, the U.S. Food and Drug Administration (FDA) hasn’t determined the cannabis plant itself to be effective for treating any particular disease or condition.
On the other hand, the FDA has approved some preparations of purified cannabinoids for therapeutic use. These are Epidiolex, which contains purified CBD and is used to treat seizures caused by Lennox-Gastaut or Dravet syndromes; Marinol and Syndros, which contain a synthetic form of THC and are used to treat anorexia in AIDS patients; and Cesamet, which contains a synthetic form of THC and is used to treat chemotherapy-induced nausea in cancer patients.
Insufficient evidence to support using cannabis to treat various diseases
Several studies show that cannabinoids can affect tumor growth; however, most of this data is based on animal studies and small clinical trials[1,2]. A recent review found that most of the published, peer-reviewed case reports provide insufficient data to support the claim for cannabis as a cancer treatment. It also reported that no clinical data support the anti-cancer benefit suggested by preclinical studies[3]. The differences between pre-clinical and clinical evidence of the effectiveness of cannabis as a potential cancer treatment are explained in this earlier claim review by Health Feedback.
There is growing interest in cannabis and its components as a possible treatment for post-traumatic stress disorder (PTSD). PTSD is a condition that develops as an adjustment to a traumatic event and can lead to significant disruption of personal and interpersonal functioning]. However, to date, there is insufficient evidence on the benefits and harms of cannabis as a treatment for PTSD. A systematic review on the benefits of cannabis for the treatment of PTSD found “very scant evidence with medium to high risk of bias” among the published reviews and observational studies[4].
Cannabis has been proposed as a possible therapy for epilepsy. Epilepsy is a neurological disorder characterized by the absence or excess of signaling between nerve cells in the brain, resulting in seizures. As stated above, the U.S. FDA approved a drug containing purified CBD to treat two rare forms of epilepsy. According to the Epilepsy Foundation, a non-governmental organization based in the U.S., research on cannabis so far has only found that CBD is helpful for treating some kinds of epilepsy.
Another proposed use of cannabis is the treatment of glaucoma. Glaucoma is a disease that can damage the optic nerve of the eye, leading to vision loss. Glaucoma is one of the leading causes of blindness in the world. Early treatment, which includes reducing intraocular pressure, can prevent severe vision loss. Clinical studies have found that cannabis lowers intraocular pressure in both healthy individuals and those with glaucoma[5]. However, this effect has a short duration (between three to four hours), which makes cannabis less effective for the treatment of glaucoma than other treatments already available.
In a 2009 statement, the American Glaucoma Society recommended against the use of cannabis for the treatment of glaucoma:
“Although marijuana can lower the intraocular pressure (IOP), its side effects and short duration of action, coupled with a lack of evidence that it use alters the course of glaucoma, preclude recommending this drug in any form for the treatment of glaucoma at the present time.”
Regarding the side effects of marijuana, the statement warned that “marijuana’s mood altering effects would prevent the patient who is using it from driving, operating heavy machinery, and functioning at maximum mental capacity”.
Finally, clinical evidence of the usefulness of cannabis for treating anxiety and depression is limited[6,7]. Some studies evaluated the efficacy of purified CBD for treating anxiety but had limitations, including short duration of studies, lack of a placebo control group and low representation of both female animals in preclinical studies and women in clinical studies[7].
Another study found a reduction in anxiety and depressive symptoms following cannabis use, but this was an observation study without a placebo control group[6]. On the other hand, there is evidence suggesting an association between cannabis use and the development of mental health disorders, including depression and schizophrenia[8], although the cause-effect relationship between cannabis use and depression is still unclear.
Overall, there isn’t clinical evidence indicating that cannabis is useful for treating cancer or other conditions. Future clinical studies with a larger number of participants and a longer duration may shed more light on this question.
For now, we only have scientific evidence showing that specific cannabinoids are useful for the treatment of a few conditions, such as drugs containing purified or synthesized forms of THC or CBD, for which the U.S. FDA has already approved. Cannabis contains a mixture of different chemicals at varying concentrations, and consuming cannabis isn’t the same as taking specified doses of purified or synthesized forms of THC or CBD, which makes the meme potentially misleading.
REFERENCES
- 1 – Wilkie et al. (2016) Medical Marijuana Use in Oncology. JAMA Oncology.
- 2 – Hinz and Ramer (2022) Cannabinoids as anticancer drugs: current status of preclinical research. British Journal of Cancer.
- 3 – Guggisberg et al. (2021) Cannabis as an Anticancer Agent: A Review of Clinical Data and Assessment of Case Reports. Cannabis and Cannabinoid Research.
- 4 – O’Neil et al. (2017) Benefits and Harms of Plant-Based Cannabis for Posttraumatic Stress Disorder. Annals of Internal Medicine.
- 5 – Green (1998) Marijuana Smoking vs Cannabinoids for Glaucoma Therapy. JAMA Ophthalmology.
- 6 – Martin et al. (2021) Antidepressant and Anxiolytic Effects of Medicinal Cannabis Use in an Observational Trial. Frontiers in Psychiatry.
- 7 – Wright et al. (2020) Use of Cannabidiol for the Treatment of Anxiety: A Short Synthesis of Pre-Clinical and Clinical Evidence. Cannabis and Cannabinoid Research.
- 8 – National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Population Health and Public Health Practice; Committee on the Health Effects of Marijuana: An Evidence Review and Research Agenda. The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. National Academies Press (U.S.)