- Health
COVID-19 vaccines aren’t expected to be available for the public in several weeks, contrary to viral claim on social media
Key takeaway
Several COVID-19 vaccines are currently being developed, but it is extremely unlikely that they will be released within three weeks. It is also unlikely that a COVID-19 vaccine will be compulsory for everyone. Even if a COVID-19 vaccine candidate successfully reaches Phase 3 of clinical trials, it must still go through a long process of approval and manufacturing.
Reviewed content
Verdict:
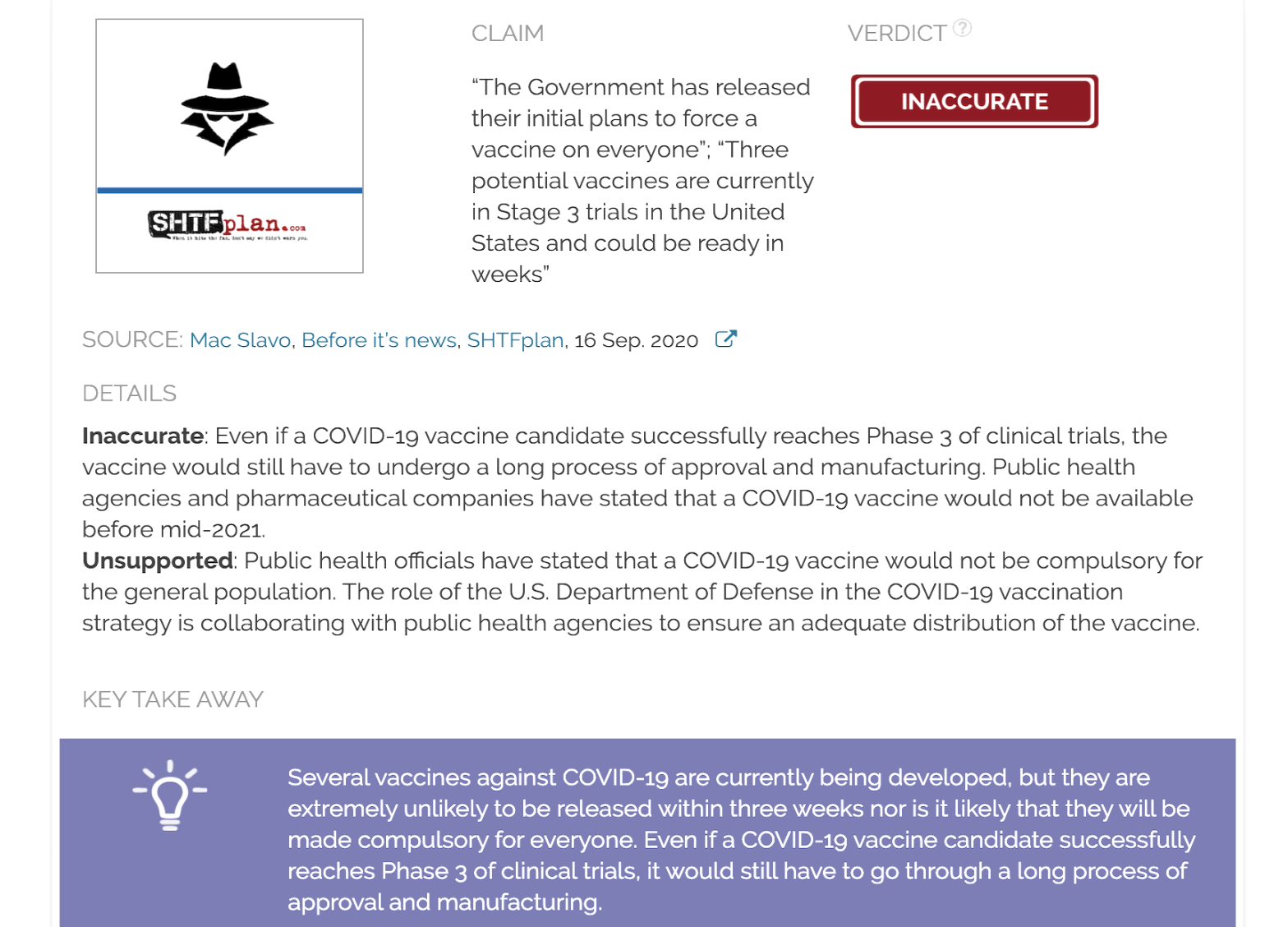
Claim:
“The Government has released their initial plans to force a vaccine on everyone”; “Three potential vaccines are currently in Stage 3 trials in the United States and could be ready in weeks”
Verdict detail
Inaccurate: Even if a COVID-19 vaccine candidate successfully reaches Phase 3 of clinical trials, the vaccine must still undergo a long process of approval and manufacturing. It is extremely unlikely that a COVID-19 vaccine will be available “in weeks” for the general public, as claimed. This also contradicts statements from public health officials and pharmaceutical companies.
Unsupported: Public health officials stated that a COVID-19 vaccine will not be compulsory for the general population. The U.S. Department of Defense is collaborating with public health agencies to ensure the adequate distribution of a COVID-19 vaccine.
Full Claim
“The Government has released their initial plans to force a vaccine on everyone”; “Three potential vaccines are currently in Stage 3 trials in the United States and could be ready in weeks”
Review
An article published by SHTFplan.com on 16 September 2020 and reposted on the website Beforeitsnews.com titled “the [U.S] government has released their initial plans to force a [COVID-19] vaccine on everyone” went viral in early October, receiving more than one million shares on Facebook alone. The article claims that the COVID-19 vaccine “could be ready in weeks” and that the military would be involved in forcing the public to be vaccinated. The claim was mostly shared by Facebook groups that oppose vaccines as well as groups that promote conspiracy theories.
The article cites a 15 September 2020 interview with the U.S. President Donald Trump on the television show “Fox and Friends” as evidence that a COVID-19 vaccine will be available in weeks. During the interview, Trump claimed that the vaccine will be ready “in a matter of weeks” and that it will be delivered “immediately upon getting it.” However, these statements contradict scientists’ estimates of the vaccine timeline.
A vaccine candidate must go through multiple stages of testing, including clinical trials and an approval process, before it is made available to the general public. Phases 1 and 2 of a clinical trial evaluate vaccine safety in a small group of volunteers and determine the most effective dose. In Phase 3, the vaccine is tested in thousands of people to study its effectiveness. If successful, manufacturers must apply for approval from the U.S. Food and Drug Administration (FDA) demonstrating that the vaccine is not only safe and effective, but also that the manufacturing process is consistent. This means that different vaccine lots must have similar safety and effectiveness to the ones used in the clinical trials. Once approved, manufacturers must produce the vaccine on a large scale. Even after approval, the FDA and the CDC continue to monitor vaccine production and safety.
Currently, three COVID-19 vaccine candidates developed by researchers in the U.S. are now in Phase 3 clinical trials. One of these vaccines, developed by Janssen and funded by the U.S National Institutes of Health (NIH), uses a weakened adenovirus responsible for the common cold. This virus carries the information to express the spike protein of SARS-CoV-2, the virus that causes COVID-19, in human cells[1]. Exposure to this protein enables our immune system to recognize the virus in future encounters and allows the immune system to mount a rapid defense against infection.
The other two vaccine candidates also rely on the same immunological principle, although they achieve the same effect through different means. These candidates use lipid nanoparticles to carry fragments of RNA material into human cells, which will then produce certain proteins of the SARS-CoV-2 virus that the immune system will identify. One of these RNA vaccine candidates is being developed by Moderna Therapeutics in collaboration with the NIH[2,3]. The other candidate was produced by an international collaboration between the pharmaceutical companies Pfizer and BioNTech[4].
Stéphane Bancel, Moderna’s CEO, explained in an interview with the Financial Times that “November 25 is the time we will have enough safety data to be able to put into an emergency use authorization file that we would send to the FDA.”
The article also makes the unsupported claim that the COVID-19 vaccine will be compulsory for everyone by the end of 2020, and that compliance by the general public will be enforced by the U.S. military. This claim may be based on a misinterpretation of a 16 September 2020 press release from the U.S. Department of Health and Human Services announcing an unprecedented collaboration between the U.S. Department of Defense and the CDC to ensure the adequate distribution of a COVID-19 vaccine.
The CDC’s Interim Playbook outlines the strategy for efficiently delivering and administering a COVID-19 vaccine to the public. According to the playbook, vaccination will start gradually among health workers, essential workers, and vulnerable groups before ramping up widespread distribution for the general public.
Ever since scientists began research on a COVID-19 vaccine, several articles and social media posts claimed without evidence that a COVID-19 vaccine will be compulsory in different countries, including the U.S. However, public health officials say mandatory vaccination is very unlikely. Anthony Fauci, director of the U.S. National Institute of Allergy and Infectious Diseases, explained during a video talk organized by George Washington University that a nationwide mandate of the COVID-19 vaccine “would be unenforceable and not appropriate.” But he also pointed out that the vaccine could be mandatory for certain groups of people, as is currently the case with flu vaccines for health workers at the NIH. Also, some states might decide to make a COVID-19 vaccine mandatory for children to attend school, similar to the measles vaccine.
In summary, the claim that a COVID-19 vaccine will be available “in weeks” for the general public is extremely unlikely, and contradicts estimates from scientists, public health officials, and vaccine manufacturers provided at the time the claim was made. This is because widespread COVID-19 vaccination requires large-scale production of the vaccine to ensure sufficient doses for the public. The claim that a COVID-19 vaccine will be mandatory for the general population is currently unsupported.
READ MORE
This article from National Geographic summarizes all COVID-19 vaccine candidates that have reached Phase 3 and explains how they work.
This review was updated to more accurately reflect the timeline of actual events in the COVID-19 vaccine approval and rollout. In September 2021, Robert Redfield, then director of the U.S. CDC, and Stéphane Bancel, Moderna’s CEO, projected that a COVID-19 vaccine wouldn’t be expected before mid-2021. But events moved much quicker than anticipated, and on 11 December 2020, the U.S Food and Drug Administration (FDA) issued the first Emergency Use Authorization for a COVID-19 vaccine, specifically the mRNA vaccine developed by Pfizer and BioNTech. Vaccination rollout in the U.S. started on 14 December 2020, beginning with groups at high risk of illness, such as healthcare workers and nursing home staff.
As such, we updated the review’s headline and removed statements by Redfield and Bancel, which overestimated the amount of time that COVID-19 vaccines would take to reach the public. The verdict of the review didn’t change, as the claim that the vaccine would be available in weeks and be compulsory is false.
REFERENCES
- 1 – Sadoff et al. (2020) Safety and immunogenicity of the Ad26.COV2.S COVID-19 vaccine candidate: interim results of a phase 1/2a, double-blind, randomized, placebo-controlled trial. medRxiv. [Note: This is a pre-print that has not yet been peer-reviewed or published in a journal at the time of this review’s publication.]
- 2 – Jackson et al (2020) An mRNA Vaccine against SARS-CoV-2 — Preliminary Report. The New England Journal of Medicine.
- 3 – Corbett et al. (2020) Evaluation of the mRNA-1273 Vaccine against SARS-CoV-2 in Nonhuman Primates. The New England Journal of Medicine.
- 4 – Mulligan et al. (2020) Phase 1/2 study of COVID-19 RNA vaccine BNT162b1 in adults. Nature.
NOTES
This fact check is available at IFCN’s 2020 US Elections FactChat #Chatbot on WhatsApp. Click here, for more.